
CATEGORIES:
BiologyChemistryConstructionCultureEcologyEconomyElectronicsFinanceGeographyHistoryInformaticsLawMathematicsMechanicsMedicineOtherPedagogyPhilosophyPhysicsPolicyPsychologySociologySportTourism
Carbohydrates - Sugars and Polysaccharides
Molecular Biochemistry I
http://www.rpi.edu/dept/bcbp/molbiochem/MBWeb/mb1/part2/sugar.htm
Contents of this page:
Introduction to carbohydrates
Monosaccharides: nomenclature, stereochemistry, & cyclic forms
Sugar derivatives
Glycosidic bonds & disaccharides
Polysaccharides - amylose, amylopectin & cellulose
Glycosaminoglycans & proteoglycans
Oligosaccharides of glycoproteins & glycolipids
Lectins
| Carbohydrates (also referred to as glycans) have the basic composition: | 
|
· Monosaccharides - simple sugars, with multiple hydroxyl groups. Based on the number of carbons (e.g., 3, 4, 5, or 6) a monosaccharide is a triose, tetrose, pentose, or hexose, etc.
· Disaccharides - two monosaccharides covalently linked
· Oligosaccharides - a few monosaccharides covalently linked.
· Polysaccharides - polymers consisting of chains of monosaccharide or disaccharide units.
Monosaccharides:
| Aldoses(e.g., glucose) have an aldehyde at one end. | Ketoses (e.g., fructose) have a keto group, usually at C #2. |

| 
|
| Nomenclature for stereoisomers: D and L designations are based on the configuration about the single asymmetric carbon in glyceraldehyde. (See also Voet & Voet, 3rd Ed, p. 73). The lower representations are Fischer Projections. | 
|
| For sugars with more than one chiral center, the D or L designation refers to the asymmetric carbon farthest from the aldehyde or keto group. Most naturally occurring sugars are D isomers. D &L sugars are mirror images of one another. They have the same name. For example, D-glucose and L-glucose are shown at right. | 
|
| Other stereoisomers have unique names, e.g., glucose, mannose, galactose, etc. The number of stereoisomers is2 n, where n is the number of asymmetric centers. The six-carbon aldoses have 4 asymmetric centers, and thus 16 stereoisomers (8 D-sugars and 8 L-sugars). See diagrams of D-aldoses in Voet & Voet on p. 357, and D-ketoses on p. 358. |
| An aldehyde can react with an alcohol to form a hemiacetal. Similarly a ketone can react with an alcohol to form a hemiketal. | 
|
| Pentoses and hexoses can cyclize, as the aldehyde or keto group reacts with a hydroxyl on one of the distal carbons. E.g., glucose forms an intra-molecular hemiacetal by reaction of the aldehyde on C1 with the hydroxyl on C5, forming a six-member pyranose ring, named after the compound pyran. See also diagrams p. 359. The representations of the cyclic sugars at right are called Haworth projections. | 
|
| Fructose can form either: · a six-member pyranose ring, by reaction of the C2 keto group with the hydroxyl on C6 · a 5-member furanose ring, by reaction of the C2 keto group with the hydroxyl on C5. | 
|
Cyclization of glucose produces a new asymmetric center at C1, with the two stereoisomers called anomers, a & b.
| Haworth projections represent the cyclic sugars as having essentially planar rings, with the OH at the anomeric C1 extending either: · below the ring (a) · above the ring (b). | 
|
Because of the tetrahedral nature of carbon bonds, the cyclic form of pyranose sugars actually assume a "chair" or "boat" configuration, depending on the sugar (diagrams p. 360).
| The representation at right reflects the chair configuration of the glucopyranose ring more accurately than the Haworth projection. The displays below use the Chime plug-in. (These structure files were produced using the program Insight II from Molecular Simulations. ) | 
|
| a-D-glucopyranose | b-D-glucopyranose |
| C O H | C O H |
Click each image, using the right mouse button to change display selections.
Change the display to sticks, to make the image easier to see.
Drag the images, using the left mouse button, to give the 2 structures the same orientation, as in the diagram above.
In orienting the molecules, look for the ring O that bridges between C1 and C5, and the OH on C6, which sticks up out of the ring.
Identify & compare the orientation of the OH on C1 of each anomer. If you select atom O1 and change the display to ball & stick, the oxygen atom of the OH on C1 will be more prominent.
| Sugar derivatives. Various derivatives of sugars exist (diagrams p. 361-363), including: Sugar alcohol - lacks an aldehyde or ketone. An example is ribitol. | 
|
| Sugar acid - the aldehyde at C1, or the hydroxyl on the terminal carbon, is oxidized to a carboxylic acid. Examples are gluconic acid and glucuronic acid. | 
|
| Amino sugar - an amino group substitutes for one of the hydroxyls. An example is glucosamine. The amino group may be acetylated. At right, the acetic acid moiety is shown in red. | 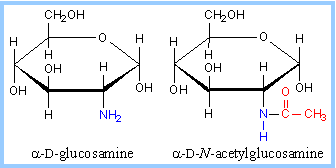
|
| N-acetylneuraminate, (N-acetylneuraminic acid, also called sialic acid) is often found as a terminal residue of oligosaccharide chains of glycoproteins. (See also p. 363.) Sialic acid imparts negative charge to glycoproteins, because its carboxyl group tends to dissociate a proton at physiological pH, as shown here. | 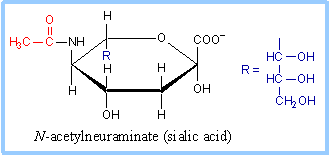
|
Glycosidic bonds: The anomeric hydroxyl group and a hydroxyl group of another sugar or some other compound can join together, splitting out water to form a glycosidic bond.
R-OH + HO-R' --> R-O-R' + H2O
| For example, methanol reacts with the anomeric hydroxyl on glucose to form methyl glucoside (methyl-glucopyranose). | 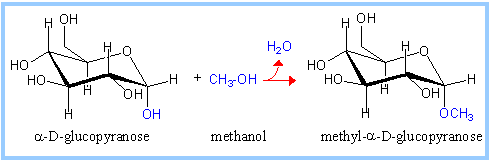
|
Disaccharides:
| Maltose, a cleavage product of starch (e.g., amylose, see below), is a disaccharide with an a(1®4) glycosidic linkage between the C1 hydroxyl of one glucose and the C4 hydroxyl of a second glucose. Maltose is the a anomer, because the O at C1 points down from the ring. | 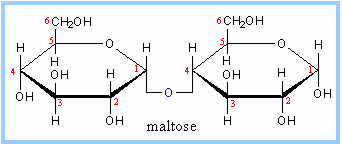
|
| Cellobiose, a product of cellulose breakdown, is the otherwise equivalent b anomer. The configuration at the anomeric C1 is b (O points up from the ring). The b(1®4) glycosidic linkage is represented as a "zig-zag" line, but one glucose residue is actually flipped over relative to the other. (See Chime view of cellulose below.) | 
|
Other disaccharides include (diagrams p. 364):
· Sucrose, common table sugar, has a glycosidic bond linking the anomeric hydroxyls of glucose and fructose. Because the configuration at the anomeric carbon of glucose is a (O points down from the ring), the linkage is designated a(1®2). The full name is a-D-glucopyranosyl-(1®2)b-D- fructopyranose.
· Lactose, milk sugar, is composed of glucose and galactose with b(1®4) linkage from the anomeric hydroxyl of galactose. Its full name is b-D-galactopyranosyl-(1®4)-a-D-glucopyranose.
Polysaccharides:
Plants store glucose as amylose or amylopectin, glucose polymers collectively called starch. Glucose storage in polymeric form minimizes osmotic effects.
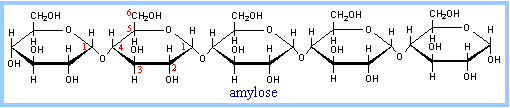
Amylose is a glucose polymer with a(1®4) glycosidic linkages, as represented above (see also diagram p. 366). The end of the polysaccharide with an anomeric carbon (C1) that is not involved in a glycosidic bond is called the reducing end.
| View the structure of amylose at right. (This data file was produced using Chem 3D, with MM2 energy minimization.) Display as sticks, and drag the image. Note the orientation of adjacent glucose residues. Compare to the diagram above. Look for the -CH2OH at C6, that sticks up out of each pyranose ring. Try other displays. You can make the O atoms more prominent by selecting atom O, and changing the display to ball & stick. Question: What is the overall shape of this polymer? | C O H |

Amylopectin is a glucose polymer with mainly a(1®4) linkages, but it also has branches formed by a(1®6) linkages. The branches are generally longer than shown above. The branches produce a compact structure, and provide multiple chain ends at which enzymatic cleavage of the polymer can occur.
Glycogen, the glucose storage polymer in animals, is similar in structure to amylopectin. But glycogen has more a(1®6) branches. See the structure of amylopectin above and diagrams on p. 367. The highly branched structure permits rapid release of glucose from glycogen stores, e.g., in muscle cells during exercise. The ability to rapidly mobilize glucose is more essential to animals than to plants.

Cellulose, a major constituent of plant cell walls, consists of long linear chains of glucose, with b(1®4)linkages. Every other glucose in cellulose is flipped over, due to the b linkages. This promotes intrachain and interchain hydrogen bonds, as well as van der Waals interactions, that cause cellulose chains to be straight and rigid, and pack with a crystalline arrangement in thick bundles called microfibrils. The regular packing of cellulose strands within a microfibril, stabilized by lateral and above/below strand interactions, is schematically represented at right.
For additional diagrams see:
| 
|
Multisubunit Cellulose Synthase complexes in the plasma membrane spin out from the cell surface microfibrils consisting of 36 parallel, interacting cellulose chains. These microfibrils are very strong. The role of cellulose is to impart strength and rigidity to plant cell walls, which can withstand high hydrostatic pressure gradients. Osmotic swelling is prevented.
A short glucose polymer, equivalent to a single cellulose strand with b(1®4)linkages, may be viewed by Chime below right. In cellulose the strand would straighter, due to interactions between adjacent strands in the cellulose fiber bundle.
| Display as sticks and drag the image. Note how each glucose residue is flipped over relative to adjacent residues, due to the b(1®4)linkages. To orient yourself, look for the ring oxygen of each residue, as well as the -CH2OH at C6, which sticks up out of each pyranose ring. (See diagram above.) You can make the O atoms prominent by selecting atom O, and changing the display to ball & stick. (This data file was produced using Chem 3D, with MM2 energy minimization.) | C O H |
| Glycosaminoglycans(mucopolysaccharides) are linear polymers of repeating disaccharides (diagrams p. 368-369). The constituent monosaccharides tend to be modified, with acidic groups, amino groups, sulfated hydroxyl and amino groups, etc. Glycosaminoglycans tend to be negatively charged, because of the prevalence of acidic groups. Hyaluronate (hyaluronan) is a glycosaminoglycan with a repeating disaccharide consisting of two glucose derivatives, glucuronate (glucuronic acid) and N-acetylglucosamine. The glycosidic linkages are b(1®3) and b(1®4). | 
|
Proteoglycans are glycosaminoglycans that are covalently linked to serine residues of specific core proteins. The glycosaminoglycan chain is synthesized by sequential addition of sugar residues to the core protein.
Some proteoglycans of the extracellular matrix bind non-covalently to hyaluronate via protein domains called link modules. For example:
- Multiple copies of the aggrecan proteoglycan associate with hyaluronate in cartilage to form large complexes. See diagrams & micrograph p. 371.
- Versican, another proteoglycan, binds hyaluronate in the extracellular matrix of loose connective tissues.
- See web sites on aggrecan and aggrecan plus versican.
| Heparan sulfate is initially synthesized on a membrane-embedded core protein as a polymer of alternatingglucuronate and N-acetylglucosamine residues. Later, in segments of the polymer, glucuronate residues may be converted to the sulfated sugar iduronic acid, while N-acetylglucosamine residues may be deacetylated and/or sulfated. | 
|
| Heparin, a soluble glycosaminoglycan found in granules of mast cells, has a structure similar to that of heparan sulfates, but is relatively highly sulfated. When released into the blood, it inhibits clot formation by interacting with the protein antithrombin. Heparin has an extended helical conformation. Charge repulsion by the many negatively charged groups may contribute to this conformation. The heparin molecule depicted at right includes 10 residues, alternating IDS (iduronate-2-sulfate) and SGN (N-sulfo-glucosamine-6-sulfate). Color key: C O N S | 
|
Some cell surfaceheparan sulfate glycosaminoglycans remain covalently linked to core proteins associated with the plasma membrane.
|  Syndecan Syndecan
|
Oligosaccharides of glycoproteins and glycolipids: Oligosaccharides that are covalently attached to proteins or to membrane lipids may be linear or branched chains. They often include modified sugars, e.g., acetylglucosamine, etc.
| O-linked oligosaccharide chains of glycoproteins vary in complexity. They link to a protein via a glycosidic bond between a sugar residue and a serine or threonine hydroxyl (diagram p. 376). O-linked oligosaccharides have roles in recognition, interaction. (See discussion of lectins below.) N-acetylglucosamine (abbreviated GlcNAc) is a common O-linked glycosylation of protein serine or threonine residues. Many cellular proteins, including enzymes and transcription factors, are regulated by reversible attachment of GlcNAc. Often attachment of GlcNAc to a protein hydroxyl group alternates with phosphorylation, with these two modifications having opposite regulatory effects (stimulation or inhibition). | 
|
| N-linked oligosaccharides of glycoproteins tend to be complex and branched. Initally N-acetylglucosamine is linked to a protein via the side-chain N of an asparagine residue in a particular 3-amino acid sequence. | 
|
| Additional monosaccharides are added, and the N-linked oligosaccharide chain is modified by removal and addition of residues, to yield a characteristic branched structure, as at right. (See also p. 376). Many proteins secreted by cells have attached N-linked oligosaccharide chains. Genetic diseases have been attributed to deficiency of particular enzymes involved in synthesizing or modifying oligosaccharide chains of these glycoproteins. Such diseases, and gene knockout studies in mice, have been used to define pathways of modification of oligosaccharide chains of glycoproteins and glycolipids. | 
|
Carbohydrate chains of plasma membrane glycoproteins and glycolipids usually face the outside of the cell. They have roles in cell-cell interaction and signaling, and in forming a protective layer on the surface of some cells.
Lectins are glycoproteins that recognize and bind to specific oligosaccharides.
Concanavalin A and wheat germ agglutinin are plant lectins that have been useful research tools (discussed p. 363).
The C-type lectin-like domainis a Ca++-binding carbohydrate recognition domain present in many animal lectins. Recognition and binding of carbohydrate moieties of glycoproteins, glycolipids, and proteoglycans by animal lectins is a factor in cell-cell recognition, adhesion of cells to the extracellular matrix, interaction of cells with chemokines and growth factors, recognition of disease-causing microorganisms, and initiation and control of inflammation. For example:
· Mannan-binding lectin (MBL) is a glycoprotein found in blood plasma. It binds cell surface carbohydrates of disease-causing microorganisms and promotes phagocytosis of these organisms as part of the immune response.
|
Date: 2015-12-17; view: 4607
| <== previous page | | | next page ==> |
| and risk warnings, and advice to | | | El Derecho Colectivo del trabajo |