
CATEGORIES:
BiologyChemistryConstructionCultureEcologyEconomyElectronicsFinanceGeographyHistoryInformaticsLawMathematicsMechanicsMedicineOtherPedagogyPhilosophyPhysicsPolicyPsychologySociologySportTourism
Magnetite Biomineralization
Heinz A. Lowenstam of the California Institute of Technology first discovered biochemically-precipitated magnetite as a capping material in the radula (tongue plate) teeth of chitons (marine mollusks of the class Polyplacophora [33]). He and his students were able to demonstrate the biological origin of this material through a variety of radioisotope tracing studies and by detailed examination of the tooth ultrastructure [34-36]. Prior to this discovery, magnetite was thought to form only in igneous or metamorphic rocks under high temperatures and pressures. In the chitons, the magnetite serves to harden the tooth caps, enabling the chitons to extract and eat endolithic algae from within the outer few millimeters of rock sub-strates. Nesson and Lowenstam [36] reported the results of detailed histological and ultrastructural examinations of magnetite formation within the radula, and noted that the process begins with an initial transport of metabolic iron to the posterior end of the radula sac. This iron is deposited as the mineral ferrihydrite within a pre-formed proteinaceous mesh [34], forming one or two distinct rows of reddish teeth. This ferrihydrite is converted rapidly to magnetite via an unknown process.
Magnetotactic bacteria were the second organisms found to contain biogenic magnetite [37, 38], a typical example of which is shown in Fig. 10.3. They precipitate individual sub-micron sized magnetite crystals within an intracellular phospholipid membrane vacuole, forming structures called “magnetosomes” [39. 40]. Chains of these magnetosomes act as simple compass needles which passively torque the bacterial cells into alignment with the earth's magnetic field, and allow them to seek the microaerophilic zone at the mud/water interface of most natural aqueous environments. These bacteria swim to the magnetic north in the northern Hemisphere [37], to the magnetic south in the southern hemisphere [41, 42], and both ways on the geomagnetic equator [43, 44]. Magnetite-bearing magnetosomes have also been found in eukaryotic magnetotactic algae, with each cell containing several thousand crystals [45]. The magnetite formation process in bacteria has an overall similarity to that in chiton teeth, as both involve deposition of a ferri-hydrite-like mineral precursor prior to magnetite formation [35, 46].
Magnetite crystals formed within these magnetosome vesicles have five main features that distinguish them from magnetites formed through geological pro-
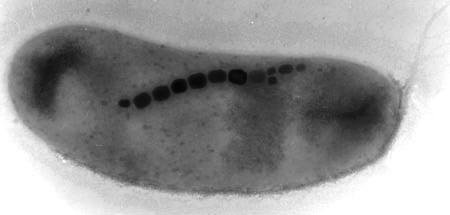
Figure 10.3. TEM image of a typical magnetotactic bacterium. The bacterium is 3 µm in size, with typical magnetite crystals on the order of 30-50 nm in length.
cesses: (1) High-resolution TEM studies reveal that bacterial magnetites are almost perfect crystals, which (2) often violate the cubic crystal symmetry of magnetite. They (3) are usually elongate in the [111] direction [40, 47-49], (4) are chemically quite pure Fe3O4, and (5) are restricted in size and shape so as to be uniformly magnetized (single-magnetic-domains). Inorganic magnetites are usually small octahedral crystals, often with lattice dislocations, chemical impurities, and other crystal defects. The elongation of biogenic crystals in the [111] direction serves to stabilize the magnetic moment of the particle, and presumably is the result of nat-ural selection for their magnetic properties [40, 50]. Bacterial magnetite crystals are restricted to a size range from 35-500 nm, with shapes that confine them to the single-domain magnetic stability field [51, 52]. Inorganic magnetites tend to have log-normal size distributions that often extend up into the multi-domain size region. Bacterial magnetites tend to be rather pure iron oxide, with no detectable titanium, chromium or aluminum, which are often present in geologically-produced magnetite. An additional feature is the alignment of the crystals into linear chains, which can be preserved in the fossil record [53, 54]. These characteristic features have enabled bacterially-precipitated magnetites to be identified in Earth sediments up to 2 billion years old [44], and possibly in 4-billion year old carbonate inclusions in the ALH84001 meteorite from Mars [55, 56].
As shown in Figures 10.4 and 10.5, many of these same features are shared by the magnetite crystals extracted from salmon [57] and from the human brain [58, 59]. The simplest interpretation of these results is that many higher organisms, including humans, possess the biochemical ability to form magnetite.
In higher animals, an obvious function for magnetite biomineralization is its role in magnetoreception [61-63]. Magnetoreception is now well established in virtually all major groups of animals [64], and specialized cells containing single-domain chains of magnetite are the best candidates for the receptor cells [60, 65]. In the brown trout, Walker et al. [60] have shown elegantly that magnetically-sensitive nerves in the ophthalmic branch of the trigeminal nerve connect to specialized, tri-lobed cells in the olfactory laminae which contain magnetite crystals. Similarly, behavioral work with honeybees and birds has shown that brief magnetic pulses are
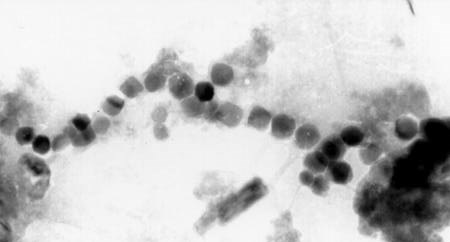
Figure 10.4. Single-domain magnetite crystals extracted from the frontal tissues of the sockeye salmon [57]. These particles are structurally nearly identical to those present in magnetotactic bacteria. Recent studies have shown that these are indeed present in the magnetosensory cells in fish [60].
able to alter the magnetic responses, confirming that a ferromagnetic material like magnetite is indeed part of the magnetic sensory system [66-72].
From an evolutionary perspective, it now seems clear that magnetite-based magnetoreception, and hence magnetite biomineralization, date back at least to the last common ancestor of Chordata, Mollusca, and Arthropoda (~600 to 900 Myr ago). The existence of magnetotactic protists argues that this genetic ability for magnetite biomineralization may go back even further to the evolution of the first eukaryotes nearly two billion years ago. Indeed, Vali & Kirschvink [40] argue that the ancestral eukaryotes probably inherited the ability to make magnetite from magnetotactic bacteria during the endosymbiotic events which formed this cell type. Therefore, the magnetite system must have been present in most of the animal phyla in the massive biomineralization episode during the Cambrian Explosion, and hence available for exaptation to form other biomineral systems. This may account for the apparent lack of a human immune response to molluscan nacre noted earlier. The fact that two of the most primitive molluscan groups, the Archaeogastropods and the Polyplacophorans, both use iron minerals to harden their radular teeth (goethite and magnetite, respectively [33, 73]) indicates that they adapted this from a preexisting iron biomineral system. Hence, magnetite biomineralization is a prime candidate for this missing “evolutionary precursor”.
Date: 2015-02-03; view: 4709
| <== previous page | | | next page ==> |
| Geological Record of the Cambrian Explosion | | | Discussion |