
CATEGORIES:
BiologyChemistryConstructionCultureEcologyEconomyElectronicsFinanceGeographyHistoryInformaticsLawMathematicsMechanicsMedicineOtherPedagogyPhilosophyPhysicsPolicyPsychologySociologySportTourism
Parts of the Written Prescription
A prescription, stripped to its barest form, consists of the superscription, the inscription, the subscription, the signa, and the name of the prescriber - written within the confines of a form. The date and patient information is followed by the superscription, which is followed by the inscription, then the subscription. Next follows the signatura.
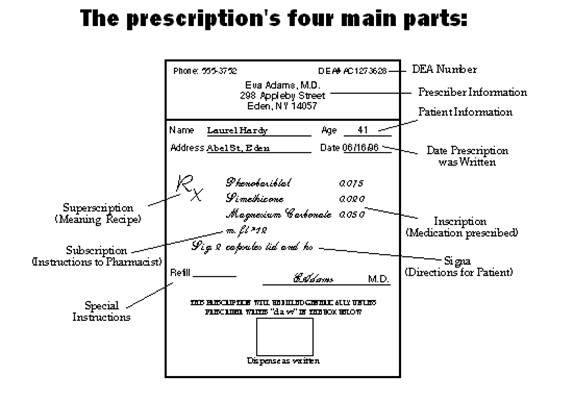
1. The superscription which consists of the heading where the symbol Rx (an abbreviation for recipe, the Latin for take thou ) is found. The RX symbol comes before the inscription.
2. The inscription is also called the body of the prescription, and provides the names and quantities of the chief ingredients of the prescription. Also in the inscription you find the dose and dosage form, such as tablet, suspension, capsule, syrup.
3. The subscription, which gives specific directions for the pharmacist on how to compound the medication. These directions to the pharmacist are usually expressed in contracted Latin or may consist of a short sentence such as: "make a solution," "mix and place into 10 capsules," or "dispense 10 tablets." However, that was in the old days. Today... doctors just name the pill!
4. The signatura (also called sig, or transcription), gives instructions to the patient on how, how much, when, and how long the drug is to be taken. These instructions are preceded by the symbol “S” or “Sig.” from the Latin, meaning "mark."
The signatura should always be written in English; however, physicians continue to insert Latin abbreviations, e.g. "1 cap t.i.d. pc," which the pharmacist translates into English when s/he prints the label, such as "take one capsule three times daily after meals". Read: Glossary of Pharmacology Terms and Symbols.
Below the Sig line is room for special instructions, such as the number of times the prescription may be refilled, if any. You will also find the purpose of the prescription, special instructions, warnings followed by the signature of the prescriber.
You should know and understand the following:
The date and patient information, which consists of the name of the party for whom it is designed and the address, usually occupies the upper part of the prescription. Sometimes age or weight is also added, though rarely.
The instruction, "take as directed" is not satisfactory and should be avoided. The directions to the patient should include a reminder of the intended purpose of the medication by including such phrases as "for pain," "for relief of headache," or "to relieve itching"
And if the patient is to receive a brand name medication, rather then generic, the physician enters NO SUBSTITUTIONS at the end of the prescription.
If there are no refills to be dispensed, it is advisable not to enter the number 0, because it can be altered by adding numbers before the zero, thus making it a 10 to receive ten refills (or more!). Always write out the word None, or No Refills!!!
The Drug Enforcement Administration (DEA) registration number system was implemented as a way to successfully track controlled substances from the time they are manufactured until the time they are dispensed to the patient.
The DEA opposes use of the DEA number for other than its intended purpose, which is tracking controlled substances, and strongly opposes insurance company practice of requiring that a DEA number be placed on prescriptions for non-controlled substances.
Not all medications require prescriptions. There are certain medications on the market that can be purchased over the counter, thus their name over-the-counter drugs (OTC.)
Date: 2014-12-29; view: 17940
| <== previous page | | | next page ==> |
| List A - narcotic and poison medicines. | | | Safety Tips |